FDA Warns Against Certain At-Home Cleaners for Your CPAP Machine
| Key Points |
|---|
|
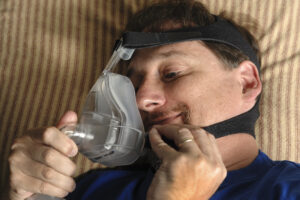
For those who struggle with obstructive sleep apnea, a continuous positive airway pressure machine (CPAP) is a game-changing piece of technology. Several companies have begun marketing at-home cleaning systems that use ozone gas or ultraviolet (UV) light. According to the FDA, however, these machines may actually worsen patients’ symptoms or put them at risk for exposure to toxic gases. These systems are not legally marketed for cleaning purposes. In fact, CPAP users typically won’t need a machine of any kind to clean a CPAP. This finding has resulted in a recent recall that affects millions of CPAP devices when cleaned improperly, and poses a threat to many users who depend on these machines for life-saving assistance.
The FDA reports that exposure to high levels of gases released by at-home cleaning products may increase the chance of respiratory infection. It also reports that UV light treatments can cause burns, eye damage, or increase the risk of skin cancer. Millions of Americans have been diagnosed with sleep apnea, which causes repeated pauses in breath during sleep due to a blocked airway in the back of the throat. In order for people with sleep apnea to fully benefit from using a CPAP, proper cleaning techniques are essential. Germs from your lungs, throat, or mouth can get into the CPAP mask or hose as you breathe in and out during sleep, or germs on your skin may transfer to the CPAP mask or hose. Equally important to cleaning your CPAP is proper reassembly and understanding the machine’s different parts.

Best CPAP Cleaning Methods
When it comes to cleaning your CPAP, manufacturers and experts tend to recommend daily cleaning — weekly cleanings are also acceptable, but as a bare minimum. There have been cases of serious illness that can be traced back to dirty CPAP machines, and CPAP users who do not regularly clean their machine may also suffer from congestion, coughs, and other indications that their respiratory system is suffering.
CPAP Cleaning Supplies
- Mild soap, preferably unscented and without moisturizing ingredients
- White vinegar (if you use a humidifier tank)
- Warm, drinking-quality water
- A sink, tub, or bucket that is large enough to hold your hose or tubing
- A clean, non-abrasive towel
Reporting A Problem
If you are an owner of a recalled CPAP machine, or have experienced issues due to improper cleaning techniques, immediate reporting of an injury or problem associated with your device can help the FDA better identify the risks associated with illegally marketed cleaning products. If you experience respiratory difficulty or other symptoms after using any UV light or ozone gas products to clean, disinfect or sanitize CPAP machines or accessories, use the MedWatch Voluntary Reporting Form to report the problem.
References
2 Sources
-
U.S. Food and Drug Administration. (2020, February 27). CPAP machine cleaning: Ozone, UV light products are not FDA approved., Retrieved July 8, 2021, from
https://www.fda.gov/consumers/consumer-updates/cpap-machine-cleaning-ozone-uv-light-products-are-not-fda-approved -
Schnirman, R., Nur, N., Bonitati, A., & Carino, G. (2017). A case of legionella pneumonia caused by home use of continuous positive airway pressure. SAGE Open Medical Case Reports, 5, 2050313X1774498.
https://pubmed.ncbi.nlm.nih.gov/29238576/