When you buy through our links, we may earn a commission. Products or services may be offered by an affiliated entity. Learn more.
What Are Micro CPAP Devices and Do They Work?
- Micro CPAP devices are being designed to offer patients with obstructive sleep apnea a less intrusive treatment option, but development is in early stages and efficacy is unproven thus far.
- The main micro CPAP prototype in development is maskless, hoseless, sits under the nostrils, and is designed as single-use.
- Micro CPAP devices are sometimes confused with other devices currently on the market that are simply smaller, travel-sized CPAP machines, but they are not the same thing.
People who have obstructive sleep apnea (OSA) are typically prescribed CPAP therapy as a first-line treatment. Although it’s proven to be the most effective when used properly, it can be challenging for patients to get used to sleeping with a mask with hose attachments that pumps air through their mouth and/or nose.
One company is working on developing a micro CPAP device that does away with hoses and masks, and delivers treatment via a small device that inserts into the nostrils. While the idea is appealing for many people struggling with CPAP treatment, the device is not yet perfected or tested, and any potential approval from the U.S. Food and Drug Administration (FDA) could be several years away.
Still, for those curious, we’ll explain what these devices are, how they work, and how they differ from traditional CPAP machines.

Exploring CPAP Machines — But Haven’t Been Tested Yet?
Start with our easy, at-home sleep test. It’s stress-free and effective. Get results within a few days.
What Is a Micro CPAP Device?
A micro CPAP device is a small, battery-operated CPAP alternative to treat OSA that’s intended to be less invasive than traditional CPAP machines, which some find cumbersome, noisy, and restrictive. Companies developing the technology are working to produce a compact design that is maskless, hoseless, and battery-powered.
To date, micro CPAP prototypes are still experimental, and thorough patient testing still needs to be done to determine efficacy. While you may see some available products marketed as “micro” CPAP or “travel CPAP” devices, those are different, as they’re simply compact versions that are FDA-approved for use when travelling. One example is the Somnetics Transcend Micro Auto-CPAP.
As for micro CPAP devices that are intended to be used without masks or hoses, there aren’t any that have received FDA approval as a treatment for OSA.
Are Micro CPAP Machines FDA-Approved?
Micro CPAP devices are not currently approved by the FDA as a treatment for sleep apnea. To ensure consumer safety, the FDA uses a rigorous process of testing and review for new medical devices. Since a working prototype doesn’t currently exist, it’s difficult to predict when or if the device will receive approval.
How Do Micro CPAP Devices Work?
The idea behind micro CPAP devices is to eliminate the need for a tabletop CPAP machine and bulky masks and hoses to deliver positive airway pressure. To do this, the company designing the technology is developing interior components called microblowers. These tiny displacement pumps are intended to create positive airway pressure by using electrostatic charge.
Similar to a bellows that fans the flames of a fire, the design sucks air in one end and forces it out the other end of each microblower. The single-use micro CPAP device is designed to be battery-powered, with an intended battery life of eight hours.
While the technology sounds promising, thus far, the product remains in early stages of development.
Do Micro CPAP Machines Work as a Treatment for OSA?
Since micro CPAP technology is still in the development stage, it’s difficult to determine whether it will be approved as a treatment for obstructive sleep apnea. Based on the information currently available, the device appears to have several shortcomings when compared with traditional CPAP machines.
Micro CPAP vs. Traditional CPAP: What’s the Difference?
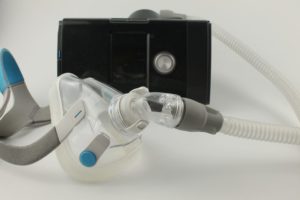
A traditional CPAP machine consists of several components:
- Tabletop unit that plugs into a wall outlet and generates continuous pressure
- Mask that fits over the sleeper’s face and delivers pressurized air to the airways
- Hose that connects the mask to the tabletop machine
To use a traditional CPAP device, sleepers must connect all the components, adjust the mask, turn on the device, and find a comfortable sleep position. Traditional CPAP machines also require regular maintenance, including changing the interior air filter and cleaning the hose and mask.
In comparison, the micro CPAP prototype features silicon nose buds that sit inside the nostrils. The body of the device rests just below the nose and contains the inhalation and exhalation vents and electronic components intended to generate positive airway pressure.
Pressure Settings
CPAP therapy requires a doctor’s prescription that includes a specific pressure setting based on the individual’s needs. CPAP pressure settings are measured in centimeters of water pressure (cm H2O). Most CPAP machines have pressure settings ranging from 4 to 20 cm H2O, though some go higher.
It’s unclear if micro CPAP devices will be able to deliver this same range of pressure. The company developing the technology claims that it will be capable of reaching more than 20 cm H2O.
However, the last technical update from the company, released in February 2020, showed that the microblowers had been demonstrated to blow air at around 5 cm H2O — far below the level needed to bring the product to market. No public updates have been provided since, leaving the status of the technology uncertain.
Pressure Control
The company designing micro CPAP technology states that the early device versions will be available in a range of pressure values so you can match the number prescribed for you. Future iterations may be able to self-adjust based on the patient’s breathing, similar to how an APAP machine works.
Humidity Control
Many people experience side effects from CPAP therapy, such as dry mouth or dry nasal passages. To mitigate these issues, some CPAP machines are equipped with humidifiers to add moisture to the pressurized air. The design of the micro CPAP prototype aims to mitigate this problem automatically without the use of humidification.
Device Disposal
The in-progress micro CPAP prototype is a single-use device. The company is open to future devices that can be recharged, and details regarding how to dispose or recycle are being worked out.
What Are the Alternatives to Micro CPAP Devices?
While micro CPAP devices won’t be on the market as an FDA-approved treatment for OSA anytime soon, there are some other options for people who struggle with traditional CPAP therapy.
- EPAP therapy (expiratory positive airway pressure) is a non-machine alternative to CPAP that uses small nasal valves to create pressure during exhalation; it doesn’t add pressure during inhalation. It’s currently considered an FDA-cleared class 2 device for OSA.
- In 2025, Zepbound (tirzepatide), first known as a weight loss drug, became the first medication approved to treat OSA by the U.S. Food and Drug Administration for adults with obesity.
- Another drug in development to treat OSA is called SynAIRgy. It has completed preliminary patient trials with promising results, and announced it will be applying for FDA approval in early 2026.

Still have questions? Ask our community!
Join our Sleep Care Community — a trusted hub of product specialists, sleep health professionals, and people just like you. Whether you’re searching for the perfect mattress or need expert sleep advice, we’ve got you covered. Get personalized guidance from the experts who know sleep best.
References7 Sources
Mwenge GB, Liistro G, Smetcoren C, Debaille C. Unveiling the hidden risks of CPAP device innovations and the necessity of patient-centric testing. J Clin Sleep Med. 2025;21(2):421-425. doi:10.5664/jcsm.11384
https://jcsm.aasm.org/doi/full/10.5664/jcsm.11384Food and Drug Administration. The Device Development Process. FDA. Published February 20, 2020.
https://www.fda.gov/patients/learn-about-drug-and-device-approvals/device-development-processHooper RG. CPAP Therapeutic Options for Obstructive Sleep Apnea. Pragmat Obs Res. 2020;11:67-76. Published 2020 Jul 13. doi:10.2147/POR.S258632
https://pmc.ncbi.nlm.nih.gov/articles/PMC7381796/Rotty, M. C., Suehs, C. M., Mallet, J. P., Martinez, C., Borel, J. C., Rabec, C., Bertelli, F., Bourdin, A., Molinari, N., & Jaffuel, D. (2021). Mask side-effects in long-term CPAP-patients impact adherence and sleepiness: The InterfaceVent real-life study. Respiratory Research, 22(1).
https://pubmed.ncbi.nlm.nih.gov/33451313/Chiang A., et al. Emerging OSA Treatment Alternatives — Side-By-Side Comparison. AASM Emerging Technology Committee.; Fall 2021. Vol. 6, No. 3.
https://aasm.org/wp-content/uploads/2021/10/Montage_Tech_Article_Supplemental_Content_21.pdfFDA Approves First Medication for Obstructive Sleep Apnea. U.S. Food and Drug Administration. Published December 20, 2024.
https://www.fda.gov/news-events/press-announcements/fda-approves-first-medication-obstructive-sleep-apneaApnimed Announces Positive Topline Results in the First Landmark Phase 3 Clinical Trial of AD109, an Investigational Once-Daily Oral Pill for Obstructive Sleep Apnea – Apnimed. Apnimed. Published May 19, 2025.
https://apnimed.com/article/ad109phase3toplineresults/